Prescription Drug Legislation and Formularies
What is a Drug Formulary?
A state workers' compensation drug formulary is a list of prescription drugs, both generic and brand name, that are usually categorized as as "pre-approved", "not-approved", or an exception that may require additional pre-approval (or authorization) before dispensing.
In addition to Drug Formularies, several states have enacted or are working through legislation on Prescription Drugs, Opioid Dispensing, Opioid Management, and Controlled Substance Management.
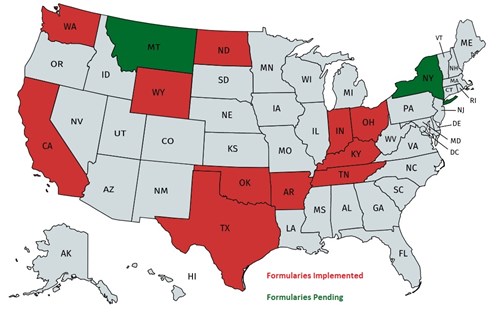
Arkansas Drug Formulary: The new drug formulary requirements are applicable to all workers’ compensations injuries on or after July 1, 2018.
For injuries that occur on or after July 1, 2018, every prescription shall be dispensed consistent with the Arkansas Workers’ Compensation Acute Drug Formulary. Any drugs not allowed on the drug formulary will require prior authorization.
The Acute Drug Formulary is a list of drugs that are designated as either “Allowed”, “Allowed up to a specific number of lifetime days” or “PA Required (Prior Authorization)”. Compound medications and drugs not listed on the formulary will require prior authorization.
- “Allowed” means the listed drug on the Arkansas Acute Drug Formulary does not require prior authorization.
- “Allowed up to a specific number of lifetime days” means the listed drug on the Arkansas Acute Drug Formulary does not require prior authorization but is only allowed for a specific number of days for the life of the claim.
- “PA Required” means the listed drug on the Arkansas Acute Drug Formulary requires prior authorization from the payer.
California Drug Formulary: The Division of Workers’ Compensation (DWC) has adopted an evidence-based drug formulary for medical providers treating injured workers beginning January 1, 2018.
The new drug formulary requirements are applicable to all workers’ compensation injuries and include the following:
- The effective date is January 1, 2018, regardless of the date of injury.
- All new injuries and claims must have a physicians’ Request for Authorization (RFA) where prescriptions are involved that are compliant to the drug formulary.
- For existing claims prior to January 1, 2018, physicians must submit a new RFA by April 1, 2018 with a treatment plan for transition to the drug formulary.
- The DWC has established a MTUS Drug List with drug designations called “Exempt and Non-Exempt”.
- RFAs with “Exempt” drug designations do NOT require Utilization Review (UR), unless there is a dispute on the body part or other compensability determinations.
- RFAs with “Non-Exempt” or unlisted drug designations will require UR.
- Prospective UR is required for physician issuance of compound drugs and physician dispensed drugs.
For additional details pertaining to the California Drug Formulary, please click on this text to view the Gallagher Bassett Client Advisory.
Indiana Drug Formulary: Indiana Senate Bill 369 was recently signed into law requiring the implementation of a workers’ compensation drug formulary based on the Official Disability Guidelines (ODG) Workers’ Compensation Drug Formulary Appendix A, effective on January 1, 2019. The drug formulary will impact all claims with an “N” drug initially dispensed on or after July 1, 2018. Rule IC 22-3-3-4.7 can be viewed in full detail on the Indiana Workers’ Compensation Board website.
The new drug formulary requirements are applicable to all workers’ compensations injuries with “N” class drug prescribed on or after 7/1/18.
Timing
- All “N” class drugs dispensed on or after 7/1/18 shall be consistent with the Indiana Workers’ Compensation drug formulary. Drugs not allowed on the formulary will require prior authorization as of 1/1/19.
- For injuries with “N” class drugs dispensed prior to 7/1/18 with continued use after 1/1/19, will be permitted until 1/1/20.
Kentucky Drug Formulary: The Kentucky Department of Workers’ Claims (DWC) has promulgated an administrative regulation (803 KAR 25:270) adopting the ODG by MCG Health pharmaceutical formulary for use in the treatment of work-related injuries and occupational disease. Drugs listed in the formulary are assigned “Y” or “N” status. Drugs assigned “Y” status may be dispensed without preauthorization and do not require utilization review. Drugs assigned “N” status, compounded drugs, and prescription drugs not listed in the formulary require preauthorization.
The formulary is available online at www.odgbymcg.com/state-formulary.
Montana Drug Formulary: The Montana Department of Labor and industry has posted Drug Formulary Regulations on their Internet Site. Please click on this text to view the regulations.
New York Drug Formulary: The New York Workers' Compensation Board (WCB) has proposed the adoption of Part 441 of 12 NYCRR and amendment of Section 440.2 of 12 NYCRR to establish a drug formulary that includes high-quality and cost-effective preauthorized medication.
The new drug formulary requirements are applicable to all workers’ compensation injuries as follows:
- On or after July 1, 2018, every new prescription shall be dispensed consistent with the Pharmacy Formulary regardless of the date of accident or disablement unless prior authorization is received to dispense a non-preferred or unlisted drug.
- On or after December 31, 2018, every refill and renewal prescription shall be dispensed consistent with the Pharmacy Formulary regardless of the date of accident or disablement unless prior authorization is received to dispense a non-preferred or unlisted drug.
The Formulary is a list of drugs that are designated as either “Preferred” or “Non-Preferred”. Additionally, some drugs are listed as second or subsequent line therapy and should only be used when the preferred drug has been deemed ineffective. Drugs not listed in the Formulary are considered “Non-Preferred”.
- “Preferred drug” means a drug on the New York Pharmacy Formulary that does not require prior authorization.
- “Non-Preferred drug” means a drug on the New York Pharmacy Formulary that requires prior authorization by the insurance carrier or self-insured employer before dispensing.
North Dakota Drug Formulary: The North Dakota Workplace Safety and Insurance has posted Drug Formulary Regulations on their Internet Site. Please click on this text to view the regulations.
Ohio Drug Formulary: The Ohio Bureau of Workers Compensation has posted its Drug Formulary Regulations on their Internet Site. Please click on this text to view the regulations.
Oklahoma Drug Formulary: The Oklahoma Workers’ Compensation Commission has posted its Drug Formulary Regulations on their Internet Site.
Please click on this text to view the regulations.
810:3-5-3. Requirements for use of closed formulary
(a) Applicability. The closed formulary adopted pursuant to 810:3-5-2 applies to all drugs that are prescribed and dispensed for outpatient use for claims with a date of injury on or after February 1, 2014.
(b) Preauthorization for claims subject to the Commission’s closed formulary. Preauthorization is only required for drugs that are excluded from the closed formulary, as defined in this Chapter.
(c) Preauthorization request. The preauthorization request must include the prescribing doctor’s drug regimen plan of care, and the anticipated dosage or range of dosages for the drugs. Failure to request preauthorization entitles an insurance carrier or employer to deny payment for the drug in question. If the insurance carrier or employer fails to respond to a preauthorization request within seventy-two (72) hours, the request shall be deemed approved.
(d) Preauthorization of intrathecal drug delivery systems:
(1) An intrathecal drug delivery system requires preauthorization and the preauthorization request must include the prescribing doctor’s drug regimen plan of care, and the anticipated dosage or range of dosages for the administration of pain medication.
(2) Refills of an intrathecal drug delivery system with drugs excluded from the closed formulary, which are billed using Healthcare Common Procedure Coding System (HCPCS) Level II J codes, require preauthorization on an annual basis. Preauthorization for these refills is also required whenever:
(A) the medications, dosage or range of dosages, or the drug regime proposed by the prescribing doctor differs from the medications, dosage or range of dosages, or drug regime previously preauthorized by that prescribing doctor; or
(B) there is a change in prescribing doctor.
(e) Treatment guidelines. Except as provided by this Subsection, the prescribing of drugs shall be in accordance with 810:3-7-1 relating to treatment guidelines. Prescription and nonprescription drugs included in the Commission’s closed formulary may be prescribed and dispensed without preauthorization.
Tennessee Drug Formulary: The Tennessee Department of Labor & Workforce Development has posted its Drug Formulary Regulations on their Internet Site. Please click on this text to view the regulations.
Texas Drug Formulary: The Texas Department of Insurance implemented a Drug Formulary on September 1, 2011. Please click on this text to view the Texas Drug Formulary Regulations.
To view Texas Department of Insurance Study on the Impact of the Texas Pharmacy Closed Formulary, please click on this text.
Washington Drug Formulary: The Washington State Department of Labor and Industry has posted its Drug Formulary Regulations on their Internet Site. Please click on this text to view the regulations.
Wyoming Drug Formulary: The Wyoming Drug Formulary has two phases, The Acute Phase is designed to treat emergent conditions (Days 0-89 from Date of Injury), while the Chronic Phase will address medication use for established claims (Day 90+from Date of Injury). The prescription medication list can be viewed by clicking on this text.
Pending Regulatory and Drug Formulary Legislation
Illinois - HB 2795 – Worker Comp Drug Formulary - Carried over to 2020 Legislative Session (selected text from source – Coventry Workers' Comp Services)
Amends the Workers' Compensation Act. Provides that the Illinois Workers' Compensation Commission, upon consultation with the Workers' Compensation Medical Fee Advisory Board, shall promulgate an evidenced-based drug formulary. Requires prescriptions in workers' compensation cases to be limited to the drugs on the formulary.
Summary for 2/14/2019 Version: This measure addresses a prescription drug formulary within the workers' compensation program. This measure stipulates, by September 1, 2020, the Workers' Compensation Commission, in consultation with the Workers' Compensation Medical Fee Advisory Board, must promulgate by rule an evidence-based drug formulary and any rules necessary for its administration. Prescriptions prescribed for workers' compensation cases shall be limited to the prescription drugs and doses on the closed formulary.
Nebraska - LB 487 - Require the Nebraska Workers' Compensation Court to adopt an evidence-based drug formulary - Carried over to 2020 Legislative Session (source – Coventry Workers' Comp Services)
Summary for 1/22/2019 Version: This measure establishes a formulary for workers' compensation drugs.
This measure permits the Nebraska Workers' Compensation Court to adopt an evidence-based drug formulary consisting of prescription drugs listed in Schedules II, III, IV and V. The formulary will apply to prescription drugs that are prescribed and dispensed for outpatient use in connection with workers' compensation claims with a date of injury on or after January 1, 2018.
A prescription drug included in the formulary adopted by the compensation court and recommended may be prescribed and dispensed without obtaining prior authorization from the workers' compensation insurer, risk management pool or self-insured employer. A prescription drug not included in the formulary adopted by the compensation court or that is included but not recommended is presumed to be reasonable if prior authorization for such drugs is obtained from the workers' compensation insurer, risk management pool or self-insured employer.
The measure requires the compensation court to consult with stakeholders regarding the adoption of a drug formulary. The stakeholders should include employers, insurers, private sector employee representatives, public sector employee representatives, treating physicians actively practicing medicine, pharmacists, and attorneys representing injured workers or employers. Any party may request a finding by an independent medical examiner if the workers' compensation insurer, risk management pool or self-insured employer denies payment for a prescription drug that is not included in the formulary adopted by the compensation court or that is included but not recommended in such formulary or if prior authorization is denied.
The compensation court may adopt and promulgate rules and regulations necessary to implement this provision.
DISCLAIMER. Thecontentof this Web site is provided for informational purposes only, from a general insurance/risk management perspective, with the understanding that Gallagher Bassett Services, Inc. (GB) is not rendering legal, accounting or other professional advice on specific matters. Accordingly, GB assumes no liability whatsoever in connection with the use of such content. GB recommends that you consult your legal counsel or other professional advisor with respect to your individual situation.
The information provided throughout this Web site is provided from a general insurance/risk management perspective and is NOT legal advice. Gallagher Bassett Services, Inc. (GB) does not provide legal advice as it is not qualified to do so. GB recommends that you seek the advice of legal counsel in order to become fully apprised of the legal implications related to the information provided above as such implications are highly dependent on the unique facts and circumstances applicable to an individual situation.
